|
3/28/2023 0 Comments Standard entropy change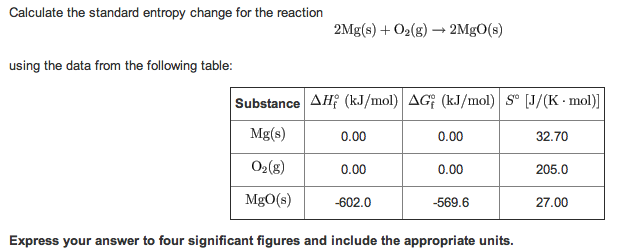
You may consult the Selected Thermodynamic Data sub-module for reference data.ģ. In the Input area, enter the problem equation then click Balance button to balance the equation then enter corresponding Entropy (S) value. Examples of Determinations of Entropy Changes in Chemical Reactions We can obtain standard entropy changes using the relationship ASe (AH e -AGe)/T. So nSo(products) nSo(reactants) The standard entropy change is equal to the sum of the standard entropies of the products minus the sum of the standard entropies of the reactants. Because Cr has a relatively high electronegativivity (1.66), Cr ions interact more. In general, the entropy change for a reaction can be determined if the standard entropies of each substance are known. Select Entropy Changes in Chemical Reactions link either from the front page (Table of Content) or from the Thermochemistry module. The change of sorption isotherms with temperature reveals the temperature dependence of the sorption equilibrium and related thermodynamic parameters, such as the standard change in Gibbs energy (G ), enthalpy (H ), and entropy (S S). 
The entropy change of the reaction can be calculated as: The n p and n r represent the moles of each product or reactant, respectively.Īn example calculating the entropy change in a reaction:įrom the Table of Thermodynamics Data, the Standard entropies of the substances involved in above reaction are: The standard entropy change of a chemical reaction can be calculated as the difference of the standard entropy values of the products and reactants. Where the first Σ to take sum of all the products in this reaction, and the second sums all the reactants. The standard entropy change for the reaction 4Fe (s) + 3O2(g)2Fe2O3(s) is -550 JK-1 at 298 K. ΔS º reaction = Σn pS º(products) - Σn rS º(reactants) Standard entropies (S°) are for one mole of substance under standard conditions (a pressure of 1 bar and a temperature of 298.15 K see details regarding standard conditions in the thermochemistry chapter of this text). With entropy, the second law of thermodynamics can be stated "In any spontaneous process there is always an increase in the entropy of the universe." The standard entropy (S º) of a substance is the value of entropy of the substance at 298 K and 1 atm.īecause entropy is a state function of a system, the entropy change in chemical reactions can be calculated by taking the difference between the standard entropy values of products and those of the reactants: 
Entropy (S) is a thermodynamic function, which can be viewed as a measure of randomness or disorder, and describes the number of arrangements (position and/or energy levels) that are available to a system existing in a given state.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
